Reinventing Lithium-ion Batteries to Reduce Costs and Boost Performance
The advanced battery market has been among the fastest growing markets. Scientists and engineers are working extremely hard to make breakthroughs in battery performance. Companies, large and small, are investing heavily on commercializing the latest battery technologies to make them cost effective.
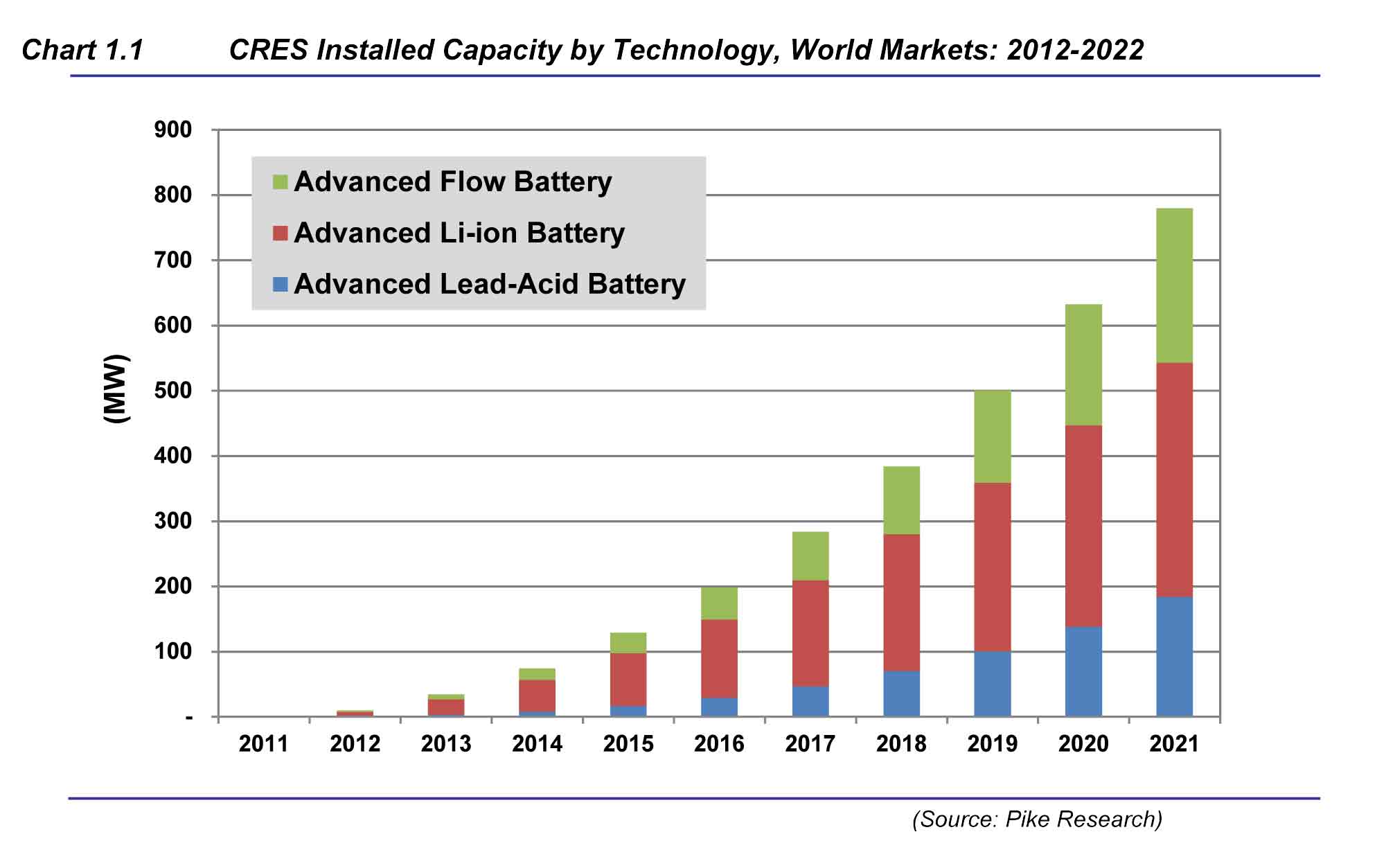
The mobile world depends on lithium-ion batteries — today's ultimate rechargeable energy store. Last year, consumers bought five billion Li-ion cells to supply power-hungry laptops, cameras, mobile phones and electric cars. “It is the best battery technology anyone has ever seen,” says George Crabtree, director of the US Joint Center for Energy Storage Research (JCESR), which is based at the Argonne National Laboratory near Chicago, Illinois. But Crabtree wants to do much, much better.

Modern Li-ion batteries hold more than twice as much energy by weight as the first commercial versions sold by Sony in 1991 — and are ten times cheaper. But they are nearing their limit. Most researchers think that improvements to Li-ion cells can squeeze in at most 30% more energy by weight (see 'Powering up'). That means that Li-ion cells will never give electric cars the 800-kilometre range of a petrol tank, or supply power-hungry smartphones with many days of juice.

Source: C.-X. Zu & H. Li Energy Environ. Sci. 4, 2614–2624 (2011)/Avicenne
Lose the dead weight
Chemical engineer Elton Cairns suspected he had tamed a promising-but-wild battery chemistry early last year, when his coin-sized cells were still going strong even after a few months of continual draining and recharging. By July, his cells at the Lawrence Berkeley National Laboratory in Berkeley, California, had cycled 1,500 times and had lost only half of their capacity1 — a performance roughly on a par with the best Li-ion batteries.
His batteries are based on lithium–sulphur (Li–S) technology, which uses extremely cheap materials and in theory can pack in five times more energy by weight than Li-ion (in practice, researchers suspect, it will probably be only twice as much). Li–S batteries were first posited 40 years ago, but researchers could not get them to survive past about 100 cycles. Now, many think that the devices are the technology closest to becoming a commercially viable successor to Li-ion.
One of Li–S's main advantages, says Cairns, is that it gets rid of the “dead weight” in a Li-ion battery. Inside a typical Li-ion cell, space is taken up by a layered graphite electrode that does little more than host lithium ions. These ions flow through a charge-carrying liquid electrolyte into a layered metal oxide electrode. As with all batteries, current is generated because electrons must flow around an outside circuit to balance the charges (see 'Radical redesigns'). To recharge the battery, a voltage is applied to reverse the electron flow, which also drives the lithium ions back.

Source Article: Nature 507, 26–28 () doi:10.1038/507026a
